Post-SSRI Sexual Dysfunction (PSSD): Understanding Persistent Sexual and Emotional Changes After SSRIs

That loss of desire, sensation, or pleasure that persisted after stopping antidepressants? It may be post-SSRI sexual dysfunction, a real but often misunderstood condition that raises big questions about symptoms, recovery, and what to do next.
If you’re here because something still feels off in your sex life long after you stopped antidepressants, you’re not imagining it: post-SSRI sexual dysfunction (PSSD) describes persistent sexual, sensory, and emotional changes that continues after stopping selective serotonin reuptake inhibitors (SSRIs), sometimes lasting months or even years.
Most people start antidepressants during a tough chapter. You were trying to feel better, more like yourself again. No one signs up for medication expecting it to create a whole new problem—especially one that affects intimacy, pleasure, and emotional connection. So when your pre-SSRI libido doesn’t return, arousal feels muted, or emotions seem strangely flat, the confusion can be intense.
You might find yourself late-night Googling things like “Lexapro sexual side effects” or asking “Is SSRI erectile dysfunction permanent?” Let’s be clear—you’re not overreacting. Many people wondering about sexual dysfunction after stopping SSRIs are told it’s caused by anxiety, depression, aging, relationship stress, or that it is “all in their head.” While some of these factors can cause sexual dysfunction, SSRI medications are increasingly recognized as another culprit. The outright dismissal of SSRIs’ role in causing persistent sexual dysfunction can be just as painful as the symptoms themselves.
I know that feeling of being lost in a medical gray zone. While I don’t personally have PSSD, I do live with chronic health issues that are poorly defined, inconsistently understood, and—at times—brushed off by doctors. It’s isolating. Overwhelming. And it often leaves you doing the exhausting work of digging through research, forums, and half-answers just to make sense of what’s happening to your own body.
While awareness of PSSD is growing, the condition is still widely misunderstood, even within healthcare. This article is here to help you get oriented. We’ll walk through what PSSD is (and what it isn’t), the symptoms people report, what the science actually shows so far, why PSSD is often missed, dismissed, or misdiagnosed, and what PSSD treatment and support options exist right now. Whether you’re searching for answers for yourself, trying to support someone you love, or looking for a clear explanation you can share with a clinician—this is a good place to start.
Healing happens through strong relationships
Compassionate, experienced mental health professionals focused on evidence-based care


.png)

What is post-SSRI sexual dysfunction (PSSD)?
Sexual side effects (aka SSRI low libido) are a well-known—and extremely common—part of taking selective serotonin reuptake inhibitors (SSRIs), the most frequently prescribed antidepressants. In fact, research suggests that nearly everyone who takes an SSRI experiences some degree of genital sensory change shortly after starting—sometimes within just 30 minutes. These changes often include reduced genital sensitivity, delayed ejaculation, muted or absent orgasms, or, less commonly, uncomfortable genital arousal or irritability.
For most people, these effects fade once they get off meds. For some, they don’t—an unknown number of people find that their sexual function doesn’t return to baseline. This condition is called post-SSRI sexual dysfunction (PSSD). It’s most commonly marked by genital numbness, weak or pleasureless orgasms, low or absent libido, and erectile dysfunction, and it can persist long after the medication is discontinued.
Despite the name, PSSD isn’t limited to SSRIs. Similar long-lasting sexual effects have also been reported with other antidepressants that affect serotonin, including serotonin-norepinephrine reuptake inhibitors (SNRIs), some anti-psychotic medications, and certain tricyclic antidepressants like amitriptyline, clomipramine, and imipramine. In other words, the issue appears to be tied to serotonin-altering medications more broadly, not just one drug class.
Even though most of us haven’t heard about PSSD, it isn’t a new issue. It’s just taken a long time to be sufficiently recognized as a condition:
- In the early 1990s, the existence of persistent sexual dysfunction after SSRI use was reported to regulators.
- In 2006, PSSD was formally described as a distinct syndrome.
- In June 2019, the European Medicines Agency (EMA) officially acknowledged that sexual dysfunction can continue even after stopping SSRIs and required manufacturers to update their drug labels accordingly.
It’s also worth mentioning that in the U.S., there is still no explicit FDA communication or SSRI labeling that warns patients that sexual dysfunction may persist after discontinuation—leaving many people unaware this outcome is even possible, says Seth Resnick, MD, DFAPA, FASAM, a board-certified psychiatrist at Radial.
And no—PSSD is not the same as depression-related sexual issues. This distinction matters. People with PSSD typically had normal sexual function before starting antidepressants. While low libido can certainly be a symptom of depression, PSSD is directly linked to antidepressant use and the complex downstream effects these medications can have on sexual response and genital sensation (we’ll get into those mechanisms later).
Finally, PSSD is about more than sex. Ongoing sexual dysfunction can deeply affect quality of life, relationships, and self-esteem. It can change how someone connects to their body and to others. That’s why understanding PSSD, validating the experience, and exploring ways to manage it truly matter.

How common is PSSD?
Short answer: “Nobody knows,” says Dr. Resnick. And that’s part of the problem. There’s no definitive data on how common post-SSRI sexual dysfunction is. While PSSD is generally thought to be rare, with one study estimating that it affects about 0.5% of people who had taken SSRIs, its true prevalence remains unknown and may be underestimated.
So, why is PSSD so hard to measure? A lot of reasons. “It is hard to quantify partly because of limitations in reporting on both ends—the patient and the clinician,” says Dr. Resnick.
Here are some of the obstacles:
- Ethical barriers to research: It would be unethical to run randomized trials that intentionally risk causing a permanent condition like PSSD.
- Imperfect measurement tools: Common sexual health questionnaires don’t assess genital sensation, a core feature of PSSD, making them ill-suited to capture the full picture.
- Lack of follow-up: Clinicians don’t routinely ask whether sexual side effects resolve after stopping antidepressants, so lingering symptoms often go unnoticed unless patients connect the dots themselves.
- Silence fueled by discomfort: Sexual concerns are still hard to talk about. Embarrassment—or a clinician’s visible discomfort—can shut the conversation down fast.
- Dismissive care experiences: Some patients report being minimized, doubted, or met with hostility when raising concerns about PSSD. Many stop seeking care altogether, removing these cases from medical records.
- Language gaps: People describe symptoms differently—some as erectile dysfunction, others as low libido or numbness—so cases may not be recognized as part of the same condition.
- Subjectivity: Erotic sensation is deeply personal and hard to quantify, making standardized assessment challenging.
Despite the gaps, emerging data offer important clues. PSSD has been reported across all ages, sexes, and ethnic groups. And while studies are limited, a few findings stand out:
- One study found that serotonergic antidepressants were linked to a more than threefold increase in the likelihood of being prescribed erectile dysfunction medication. In that study, 0.46% (about 1 in 216) of patients developed PSSD.
- In a study of 76 former antidepressant users, 52.6% reported persistent sexual dysfunction, and 26.3% experienced genital anesthesia and/or nipple insensitivity.
The big picture: we need better data and better listening. More rigorous research is essential to understand who’s at risk, how often PSSD occurs, and how best to prevent and treat it. Until then, people deserve transparent conversations about potential risks, and those experiencing PSSD deserve to be taken seriously, not brushed aside.
What are PSSD symptoms?
For many people, PSSD feels less like a “side effect” and more like a switch that didn’t turn back on. For those who experience it, the effects can be physically and psychologically debilitating. PSSD symptoms vary widely from person to person, yet clear patterns tend to emerge.
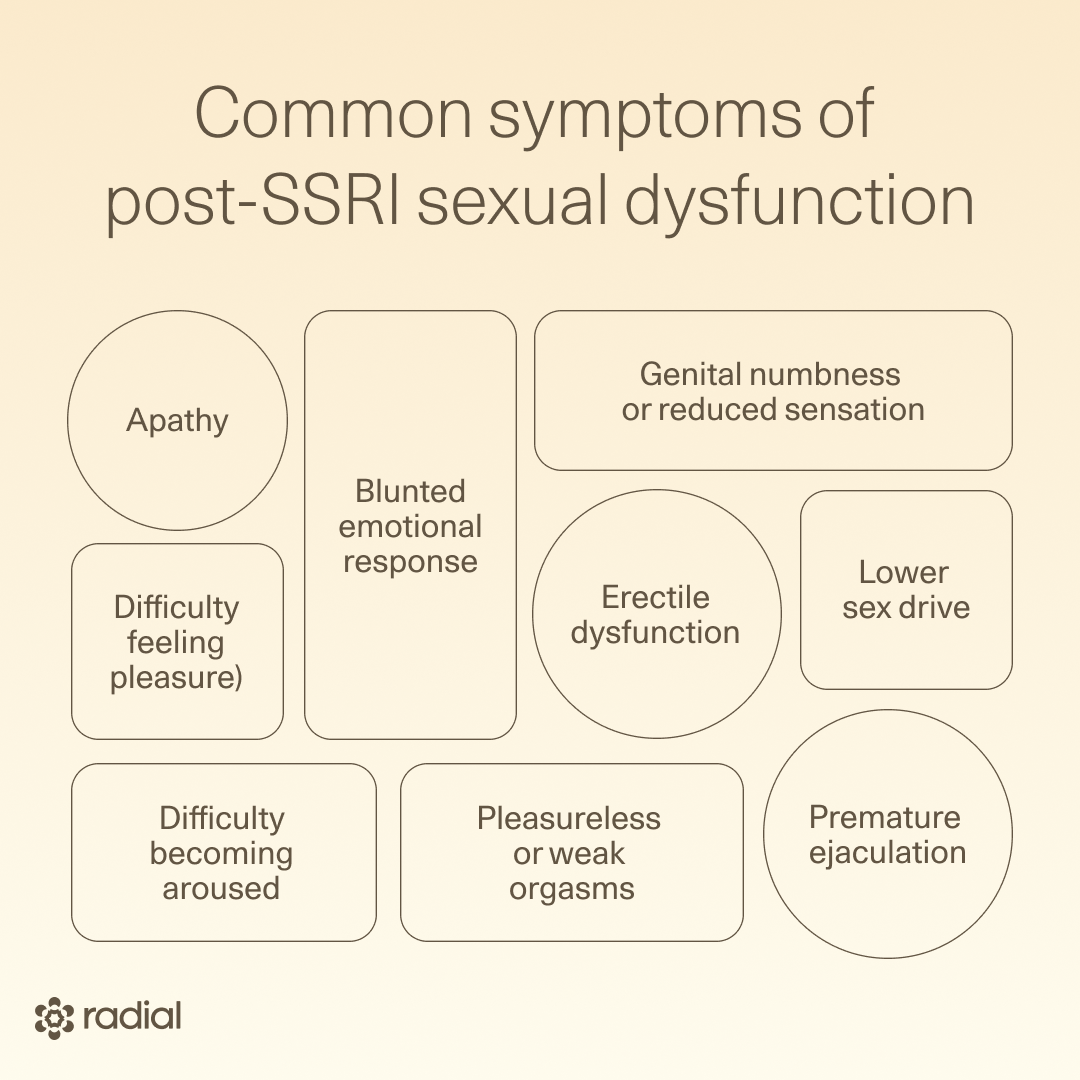
A common theme: Many people describe PSSD as a disconnect between the brain and the body or feeling cut off from genital sensation or sexual response—as if desire, arousal, or pleasure no longer translate physically. Reported sexual symptoms may include:
- Genital numbness or reduced sensation
- Decreased sex drive (low or absent libido)
- Erectile dysfunction
- Difficulty becoming aroused or reaching orgasm
- Pleasureless or weak orgasms
- Premature ejaculation
But PSSD isn’t only about sex. Growing evidence suggests that some people also experience broader emotional and motivational changes, including:
- Anhedonia (difficulty feeling pleasure)
- Apathy
- Blunted emotional response
These non-sexual symptoms can deepen the sense of disconnection and make PSSD feel like a whole-body experience, not just a sexual one.
And the ripple effects can be life-altering. For some, PSSD significantly affects relationships, self-esteem, mental health, and overall quality of life. Researchers have documented cases linked to relationship breakdowns, job loss, and, in severe situations, suicidality. That said, experiences aren’t universal—some people report little distress, unconcerned by a reduced interest in sexual activity or pleasure.
The symptom timeline is equally as unique. Symptoms of PSSD can show up after just a few doses or only become noticeable after years of use. Duration also varies widely. Some studies show SSRI-related sexual dysfunction lasting six months after discontinuation, while others suggest PSSD can persist for many years, even in people whose depression or anxiety has fully resolved.
That said, long-lasting doesn’t automatically mean permanent. While some people report symptoms that linger for years, the brain has an amazing ability to adapt and change over time. “I do believe in neuroplasticity,” says Dr. Resnick. “The brain is not just a lump of squishy tissue and a tangle of billions of neurons. It is a living, breathing, and evolving organ—and it’s this natural changeability that is its wonder.”
There’s real-world evidence to support this idea, too. In some cases, people experience “spontaneous recovery” over several years. Others report brief windows of remission—days where sensation, desire, or pleasure partially returns. Taken together, these patterns could suggest that PSSD symptoms may not stem from irreversible damage, but from changes in brain signaling that, at least for some people, can slowly shift again with time.
Why is PSSD often missed or dismissed?
PSSD regularly slips through the cracks because awareness is low among patients and healthcare professionals. If you don’t know it exists, you won’t connect the dots.
Many former SSRI users don’t realize their sexual symptoms could be linked to past antidepressant use. Instead, changes in libido, sensation, or arousal often get chalked up to aging, stress, relationship issues, or “just how things are now.” Without the concept of PSSD on the radar, the connection simply never gets made.
Providers may miss it for the same reason—and then some. Clinicians are often taught that once a drug is out of the body, its side effects should be gone too. So when sexual symptoms persist, they may assume the cause must be psychological. That assumption—while common—isn’t always accurate and can shut down further investigation.
Plus, uncertainty makes people uncomfortable (even doctors). “Doctors will steer clear from areas where they do not have the answer or feel that they cannot help with a definitive treatment or cure,” says Dr. Resnick. PSSD fits that category. Add in the pressure for providers to “have all the answers,” and it can feel safer to say, “That’s not possible,” than to sit with the unknown and explore it with curiosity, he explains.
That dismissal has real consequences. When patients feel minimized or invalidated, many stop speaking up or stop seeking care altogether. Even well-intentioned providers can struggle here, especially because PSSD symptoms overlap with depression itself, making it harder to tease apart cause and effect.
Early exposure to SSRIs muddies the waters even more. Animal studies show lasting sexual changes in rodents exposed to antidepressants early in life, and emerging human research hints at similar patterns. One study of women suggests SSRI use during childhood may disrupt the development of sexual reward systems, potentially increasing the risk of sexual desire issues later on. The diagnostic challenge? These individuals may not have a sexual “baseline” for comparison, and they’re unlikely to connect adult symptoms to antidepressant exposure years earlier.
The biggest problem of all: “There isn’t even agreement on the diagnostic criteria for PSSD,” says Dr. Resnick. Without a shared definition, how can something be reliably diagnosed or even universally acknowledged? That lack of consensus also fuels wildly inconsistent estimates, ranging from claims that nearly everyone on SSRIs experiences sexual dysfunction at some point to skepticism that persistent symptoms exist at all, he says.
So where does that leave patients? While clearer diagnostic standards and broader awareness are essential, what matters most right now is thorough evaluation. That means providers taking symptoms seriously and doing a comprehensive workup—covering physical health, sexual history, medication exposure, and ruling out other contributors like diabetes, hypertension, depression, smoking, alcohol, or substance use.
PSSD may be poorly defined, but your symptoms are real, and they deserve careful, curious, and compassionate attention.
What causes PSSD?
If you’re looking for a single, tidy answer, apologies in advance. Post-SSRI sexual dysfunction is still poorly understood. The causes? Yep, “we don’t really know that either,” says Dr. Resnick. What researchers do agree on is that PSSD is likely “complex and multifactorial,” meaning several biological processes may interact to produce symptoms, he says.
Possible risk factors researchers are exploring include: prior exposure to certain medications, genetic vulnerability, psychological stress, atypical chemical responses to antidepressants, and pre-existing conditions that affect neuroplasticity. None of these are definitive, but they help frame the leading theories.
Below are the main mechanisms scientists are actively investigating.
Serotonin receptors that stop responding via epigenetic changes
One leading theory focuses on 5-HT1A receptors, serotonin receptors that help regulate how much serotonin is released between nerve cells.
Under normal conditions, these receptors act like brakes. But with long-term SSRI use—and chronic high serotonin exposure—those brakes may loosen, says Dr. Resnick. “By taking the drug persistently, we are tricking … our nervous system, and, more specifically, the cells within it, to think that this is … the new normal,” he explains. The system adapts by downregulating these receptors, making them less sensitive.
Over time, this recalibration may allow serotonin to flood synapses more freely, says Dr. Resnick. That matters because 5-HT1A receptors also play a role in sexual motivation. Research suggests that persistent receptor desensitization—possibly driven by epigenetic changes—may continue even after the medication is stopped.
These long-lasting changes in gene expression have been observed in several brain regions tied to mood, reward, and sexual function, including the frontal cortex, hippocampus, and caudate-putamen.
Numbed peripheral nerve function
PSSD is not “all in your head”. While SSRIs act on the brain, about 95% of serotonin receptors live outside it, in the peripheral nervous system. “Yes, the nervous system extends well beyond the brain to virtually all parts of the body,” says Dr. Resnick. That includes nerves involved in sexual sensation and function.
Herein lies the potential problem: SSRIs don’t target serotonin selectively, says Dr. Resnick. They act broadly across serotonergic synapses throughout the body. One theory suggests SSRIs can trigger PSSD by flooding serotonin levels in peripheral nerves. In this scenario, excessive serotonin may “lead to an overabundance of stimulation and … may cause excitotoxicity [aka nerve cell death] and nerve damage in the peripheral nervous system,” he explains.
If this happens in genital tissue, it could help explain symptoms like genital numbness, reduced sensation, or erectile dysfunction, adds Dr. Resnick.

Neurosteroid imbalance
SSRIs are known to increase production of allopregnanolone, a neurosteroid involved in mood regulation and sexual function, says Dr. Resnick. On the surface, that sounds beneficial.
So what’s the problem? Chronic exposure may again trigger downregulation— SSRIs may “disrupt the natural function of allopregnanolone when chronically administered, causing a decline in its production and/ or altered receptor sensitivity,” explains Dr. Resnick. It’s the same adaptation issue we see with serotonin receptors like 5HT1A, he says.
Because allopregnanolone is “crucial for normal sexual function,” according to Dr. Resnick, any disturbance can have an impact, possibly contributing to PSSD.
Other hormonal changes
Another theory zooms out to the hormonal ripple effects SSRIs may cause. These include increases in serotonin and prolactin, decreases in dopamine, testosterone, oxytocin, and nitric oxide, and changes in adrenergic signaling—all of which play roles in sexual function.
A key focus is the serotonin–dopamine balance. Serotonin can inhibit dopamine release through its action on 5-HT2 receptors, says Dr. Resnick. While dopamine is often oversimplified as the “pleasure chemical,” it is crucial for sexual arousal.
When serotonin dampens dopamine signaling in reward pathways like the ventral tegmental area, it may reduce “motivation, pleasure, and sexual function,” says Dr. Resnick.
Researchers are also studying serotonin’s effects on proopiomelanocortins (hormone precursors) and melanocortins (peptide hormones), which are involved in sexual behavior. SSRIs may disrupt signaling between proopiomelanocortin and melanocortin receptors, potentially contributing to persistent dysfunction.
Relief within reach
Care covered by your insurance
Radial provides advanced mental health treatment, covered by the insurance you already use.








What PSSD treatments are available?
There’s no silver bullet, yet. The frustrating reality is that no consistently effective treatment for post-SSRI sexual dysfunction (PSSD) has been established. Well-designed clinical trials are scarce, which means there’s no clear consensus on what works, for whom, or why.
The hopeful part? Awareness of PSSD is growing—and with it, experimental approaches that may help certain symptoms in certain people. Here’s what’s currently on the radar.
Light therapy (phototherapy)
Low-power laser irradiation (also called phototherapy) is being explored as a potential treatment for symptoms of PSSD, particularly genital numbness.
In one case study, phototherapy directed at the scrotal skin and penile shaft improved penile sensitivity by about 40% in a man with PSSD-related penile anesthesia. Researchers hypothesized that the treatment helped restore the function of transient receptor potential channels—one proposed mechanism behind PSSD.
That said, the benefits were partial. While sensation and temperature sensitivity improved, symptoms like erectile dysfunction and anejaculation (i.e., inability to ejaculate during orgasm) did not. Bottom line: phototherapy may help sensory changes, but it’s far from a comprehensive fix.
Transcranial magnetic stimulation (TMS)
Repetitive transcranial magnetic stimulation (TMS) is a noninvasive treatment that uses magnetic pulses to stimulate specific brain regions involved in mood regulation. It’s FDA-cleared for treatment-resistant depression and may hold promise for PSSD, too.
In a small study using theta-burst stimulation (TBS)—a form of TMS using short bursts of stimulation that targets the dorsolateral prefrontal cortex (DLPFC)—all three participants managing bipolar disorder showed improvements in sexual dysfunction. Participants also reported increased confidence during sex and greater engagement in their relationships.
Why the DLPFC? This brain region is a “major hub” in sexual behavior and arousal circuits. It both modulates sexual response and, paradoxically, may inhibit arousal in some contexts. Because it’s close to the surface of the brain, it’s an “ideal target for neuromodulation,” but that dual role also means the effects of stimulation are complex.
Translation: TMS is promising for improving sexual dysfunction in depressed adults; however there is no data on if and how it might help people with PSSD.
Dopamine agonists
Dopamine plays a central role in sexual desire, arousal, and motivation. Drugs called dopamine agonists—designed to mimic dopamine’s effects—have been explored as potential treatments for sexual dysfunction, including PSSD.
Some evidence suggests that targeting specific dopamine receptors (notably D4 receptors) may improve erectile dysfunction in rats. However, trials using dopamine agonists like pramipexole and cabergoline for PSSD have shown little benefit overall.
Research on dopamine agonists in women is virtually non-existent. One study found that aripiprazole—an antipsychotic that partially activates dopamine D2 receptors—actually caused hypersexuality in two women with schizophrenia, possibly by stimulating dopamine pathways in the brain’s reward center (the nucleus accumbens).
But dopamine agonists come with real caveats. At higher doses, they’ve been linked to pathological gambling, compulsive behaviors, and hypersexuality. While increased libido might sound appealing on paper, these effects can cause serious distress and relationship problems.
Supplements
Accessible? Yes. Proven? Not exactly.
Some supplements are thought to support libido or genital arousal—particularly in men—and are sometimes used as alternatives for managing sexual dysfunction related to antidepressants.
One supplement, EDOVIS, has been studied indirectly in men with PSSD, showing potential benefits. It contains a blend of L-citrulline, tribulus terrestris, maca, damiana, muira puama, and folic acid—ingredients traditionally associated with male sexual function.
Not all supplements have been tested specifically for PSSD but some of those below may help aspects of sexual dysfunction. These include:
- Saffron, which may improve arousal and lubrication and reduce SSRI-related sexual side effects
- Turmeric, which may support sexual function indirectly by reducing inflammation and increasing brain-derived neurotrophic factor (BDNF), a protein that plays an important role in neuronal growth
- Ginseng, rich in ginsenosides linked to sexual performance in men and menopausal women
- L-arginine, a nitric oxide precursor that may support erectile function and genital sensation
- Tongkat Ali, associated with increased testosterone
- Horny goat weed, containing icariin, traditionally used for erectile dysfunction
- Zinc, which may support erectile function
That said, supplements aren’t harmless. Excessive or accidental overuse has been linked to nausea, vomiting, diarrhea, abdominal pain, or worse and can interact with other medications. They’re also not FDA-regulated, meaning purity, dosing, and safety aren’t guaranteed.
Rule of thumb: Always talk to a healthcare provider first, and look for third-party tested products (NSF, USP, Informed Sport) to reduce risk.
Prevention: stopping PSSD before it starts
Right now, prevention may be the most effective strategy we have. With no proven treatment for established PSSD, early intervention matters. If you’re taking an SSRI or another serotonergic antidepressant and notice sexual side effects, it’s critical to speak up early.
In some cases, discontinuing the SSRI and switching to other antidepressants that don't affect libido—under medical supervision—may reduce the risk of long-term sexual dysfunction.
Here’s what the evidence suggests so far:
- In one study, only 4% of people who switched to a dopaminergic antidepressant (amineptine) had persistent sexual dysfunction six months after stopping treatment versus 55% of patients who stayed on SSRIs who reported persistent symptoms.
- Bupropion, which has no serotonergic activity, is another promising alternative. In a study comparing bupropion with an SSRI (sertraline, aka Zoloft), patients on sertraline reported significantly more sexual dysfunction, while those on bupropion reported higher sexual satisfaction and fewer issues with desire and orgasm.
- Bupropion has also shown benefit as an add-on treatment for SSRI-related sexual dysfunction.
More of the story: If sexual side effects show up, don’t wait them out. Early medication changes may help prevent longer-term problems.
What are PSSD symptom management and support options?
There may not be a cure yet, but you’re not powerless. While a definitive treatment for PSSD remains elusive and research is moving slower than anyone would like, there are meaningful ways to manage symptoms, protect your mental health, and feel less alone as you navigate this condition.
Find a provider who believes you
Many people with PSSD report being minimized, doubted, or brushed off when they bring up symptoms. Often, this isn’t malice—it’s lack of knowledge. Unfortunately, those experiences can be so discouraging that some people stop seeking care altogether, which only deepens the isolation.
If you’ve had a frustrating experience, don’t let that be the end of the road. The most helpful providers tend to be, in Dr. Resnick’s words, “modest and curious.” They listen first, believe their patients, and resist the reflex to say “that’s not possible” just because something hasn’t been fully explained yet. As he notes, clinging only to what’s already proven can feel safe, but it leaves patients stranded when medicine doesn’t yet have all the answers.
Equally important is finding a provider who treats you as a partner, not a problem to be solved. A doctor should act “as a guide” rather than a gatekeeper, Dr. Resnick says. The best care happens when clinicians and patients bring “co-equal expertise” to the table—medical knowledge on one side, lived experience on the other, he says.
Real talk: finding the right provider can take time. But having someone who believes you and is willing to explore options with you can make a huge difference.
Get active in the PSSD community
PSSD is isolating, but you don’t have to do this alone. Even without official prevalence numbers, one thing is clear: many people are living with PSSD, and connection matters. Talking with others who truly “get it” can be deeply validating, reduce stigma, and help you feel less alone in your experience.
Peer support also offers something uniquely powerful: real-world coping strategies. Hearing how others manage symptoms, advocate for themselves, or simply get through hard days can be grounding and sometimes motivating.
Not sure where to start? Here are a few trusted spaces:
- PSSD Network–Focused on raising awareness, accelerating research, and supporting people with PSSD and their loved ones. They also offer volunteer opportunities, which many find empowering and purpose-giving.
- SIDEFXHUB (Side Effects Hub)–Dedicated to improving quality of life for people with PSSD through research, advocacy, and support. They also host WhatsApp peer support groups for direct connection.
- PSSD-focused Reddit communities–A place to read firsthand experiences, learn what has (or hasn’t) helped others, and find a sense of shared understanding.
Speak to a therapist
“Sexual health is mental health,” says Dr. Resnick. Full stop. Sexuality is deeply tied to who we are. When something disrupts it, the impact can ripple into mood, relationships, and self-esteem.
In other words, PSSD doesn’t just affect the body; it can shake your sense of identity, confidence, and emotional wellbeing. Therapy can offer critical support for both individuals with PSSD and their partners.
As Dr. Resnick puts it, a skilled mental health professional can provide “sensitivity, care, and sacred space”—listening without judgment and helping you process what you’re going through.
Certain types of therapy may be especially helpful. Cognitive-behavioral therapy (CBT) can help people with PSSD challenge unhelpful thought patterns, such as feelings of sexual inadequacy or shame, and develop healthier ways to cope with ongoing symptoms.
Communicate with your partner
PSSD doesn’t happen in a vacuum—it affects relationships, too. For couples, PSSD can feel like an earthquake, with emotional aftershocks on both sides. Partners are often deeply affected, even if they don’t experience the symptoms firsthand. That’s why open communication—and education about PSSD—is essential.
Therapy can be especially valuable here. Options include:
- CBT, to help both partners work through negative beliefs, frustration, and lowered self-esteem
- Sex therapy or couples counseling, which can help partners understand that sexual changes are a medication-related condition—not a loss of desire or attraction—and explore new ways to maintain intimacy and connection
When partners feel informed and included, it can ease misunderstandings and reduce blame on both sides.
The bottom line
PSSD can feel isolating, confusing, and deeply frustrating, especially when answers are slow and certainty is in short supply. But the growing recognition of post-SSRI sexual dysfunction is shifting the conversation. We’re learning more about how it shows up, why it’s often overlooked, and how recovery may still be possible thanks to the brain’s ability to adapt and change.
If you’re dealing with lingering sexual or emotional side effects after antidepressants, you don’t have to figure it out alone. Speaking up early, seeking out informed and curious clinicians, and advocating for yourself are powerful first steps.
Radial can help. Radial connects you with licensed clinicians—virtually or in person—who take your concerns seriously and work with you to build a personalized plan using the latest fast-acting, evidence-based treatments. If something doesn’t feel right, trust that instinct and take the next step toward answers and support.
Key takeaways
- PSSD is real and often missed. Post-SSRI sexual dysfunction can persist after stopping antidepressants, but low awareness and unclear diagnostic criteria mean it’s frequently dismissed or misdiagnosed.
- Symptoms go beyond sex. Genital numbness, low libido, and orgasm changes are common, but emotional blunting, apathy, and anhedonia can also be part of the picture.
- There’s no single cure, yet. No treatment works consistently for everyone, though options like neuromodulation, medication strategies, supplements, and symptom support may help some people.
- Support matters. Finding a provider who believes you, connecting with the PSSD community, and addressing mental and relational health can make a meaningful difference.
Frequently asked questions (FAQs)
Does PSSD ever go away?
Sometimes, but not always. Some people experience improvement over time, while others have symptoms that persist for months or years after stopping an SSRI. There’s no reliable way to predict who will recover, how long it will take, or which symptoms may improve. That uncertainty is one of the hardest parts of PSSD, and a major reason more research is urgently needed.
What are the symptoms of PSSD?
PSSD affects sexual function, sensation, and desire, and it can look different for everyone.
Common symptoms include:
- Reduced or absent libido
- Genital numbness or reduced sensation
- Difficulty becoming aroused
- Erectile dysfunction
- Delayed orgasm, weak orgasm, or inability to orgasm
- Reduced sexual pleasure or emotional “flatness” related to sex
For some people, symptoms extend beyond sex and include blunted emotions, reduced motivation, or difficulty feeling pleasure more broadly.
How common is post-SSRI sexual dysfunction?
We don’t know for sure, and it’s likely underreported. Sexual side effects during SSRI use are very common, with some data estimating 26–65% of users experience some form of sexual dysfunction. PSSD may be less common, but exact numbers are unknown, possibly due to underdiagnosis, lack of awareness, and limited long-term studies.
Which SSRI is most likely to cause PSSD?
PSSD has been reported after many SSRIs and other serotonergic antidepressants. That said, paroxetine (Paxil) is often linked to higher rates of sexual side effects during treatment.
Looking for an SSRI with the least sexual side effects? We simply don’t have the data. Truth is, PSSD can technically occur with any SSRI—and even after short-term use—so no drug is completely risk-free. If you feel you need medication to help treat your depression, buproprion (an antidepressant that doesn't work on serotonin) may be an appropriate treatment option.
Deep dive recommendations:
- NEI Podcast. E250 - Sexual Dysfunction: The Least Talked about Side Effect with Anita Clayton, MD. https://neiglobal.libsyn.com/e250-sexual-dysfunction-the-least-talked-about-side-effect-with-anita-clayton-md
Editorial Standards
At Radial, we believe better health starts with trusted information. Our mission is to empower readers with accurate, accessible, and compassionate content rooted in evidence-based research and reviewed by qualified medical professionals. We’re committed to ensuring the quality and trustworthiness of our content and editorial process–and providing information that is up-to-date, accurate, and relies on evidence-based research and peer-reviewed journals. Learn more about our editorial process.
Let's connect
Get started with finding the right treatment for you or someone you care about
Get started




